Importance of supply chain in medical device manufacturing?

The supply chain is a critical component of medical device manufacturing, impacting everything from production timelines and cost management to product quality, regulatory compliance, and customer satisfaction. The importance of an efficient and resilient supply chain in medical device manufacturing can be summarized across several key areas:
1. Timely Delivery of Quality Products:
- On-time Production: A well-managed supply chain ensures that materials, components, and finished goods are delivered at the right time, avoiding production delays that could disrupt the availability of medical devices.
- Regulatory Compliance: Many medical devices are subject to strict regulatory standards (e.g., FDA, EU MDR, CDSCO). A reliable supply chain ensures compliance with quality assurance protocols and certifications, as regulatory bodies often require documented traceability of materials from suppliers to end products.
2. Cost Management:
- Cost Efficiency: Efficient supply chain management helps reduce the cost of goods sold (COGS) through better sourcing, transportation, and inventory management practices. By optimizing procurement, medical device manufacturers can reduce production costs and improve profit margins.
- Minimizing Stockouts and Overstocks: Supply chain management plays a key role in inventory control, helping to strike the right balance between having enough materials on hand and avoiding excess inventory, which ties up capital and incurs storage costs.
3. Product Quality Assurance:
- Quality of Raw Materials and Components: The supply chain ensures that high-quality raw materials and components (such as plastics, metals, sensors, and electronics) are sourced from reliable suppliers who meet the required industry standards.
- Traceability: The supply chain provides the traceability of materials, components, and finished products. In case of a product recall, knowing the exact source of the materials or parts used in the device is crucial for safety, legal, and ethical reasons.
4. Regulatory Compliance and Documentation:
- Documentation for Certifications: Medical device manufacturers must maintain extensive documentation to comply with regulatory requirements. This includes maintaining a detailed record of supplier certificates, raw material specifications, test results, and quality control checks.
- International Standards: In countries like the U.S. and EU, medical device manufacturers must demonstrate compliance with Good Manufacturing Practices (GMP), ISO 13485, and other international standards. A well-managed supply chain ensures that materials meet these standards and are appropriately certified.
5. Risk Mitigation:
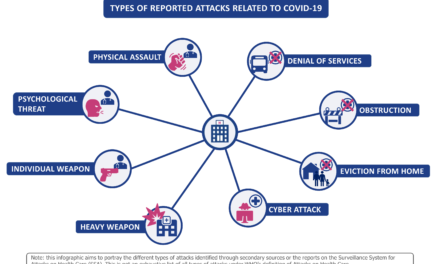
- Disruption Management: The COVID-19 pandemic and other global disruptions highlighted the vulnerability of the medical device supply chain. Effective risk management within the supply chain (e.g., diversification of suppliers, local sourcing, and contingency planning) is essential to safeguard against global supply chain disruptions.
- Supply Chain Resilience: By having multiple suppliers for critical components, manufacturers can reduce the risk of sudden shortages due to factors like natural disasters, geopolitical instability, or pandemics.
6. Compliance with Ethical Standards:
- Ethical Sourcing: In the medical device industry, ethical considerations extend beyond the quality of materials. Manufacturers are under pressure to ensure that suppliers comply with ethical labor practices, environmental sustainability, and responsible sourcing.
- Sustainability: As demand for more sustainable products grows, medical device companies are increasingly expected to source eco-friendly materials, minimize waste, and ensure that their supply chain practices are environmentally responsible.
7. Inventory Management and Lean Manufacturing:
- Lean Manufacturing: Supply chain management plays a key role in lean manufacturing by reducing waste and ensuring the efficient use of resources. It helps streamline production workflows by maintaining optimal inventory levels, reducing overproduction, and excessive stock.
- JIT (Just-in-Time) Inventory: Medical device companies can adopt Just-in-Time inventory strategies to avoid overstocking while still ensuring a smooth production process.
8. Global Sourcing and Supply Chain Diversification:
- Global Sourcing: Many medical device manufacturers source parts from multiple countries due to cost advantages, availability of specialized components, and technological expertise. However, this can add complexity to the supply chain, requiring rigorous quality controls and coordination.
- Diversification: To mitigate risk, manufacturers diversify their suppliers across different geographies and companies, avoiding over-dependence on a single source or region for critical components.
9. Supplier Relationship Management:
- Collaborative Relationships: Building strong relationships with suppliers ensures consistent quality, timely deliveries, and collaboration in the development of new components. These partnerships help manufacturers align their supply chain with their production needs and innovation goals.
- Supplier Audits: Regular audits of suppliers are crucial for ensuring that they maintain the necessary quality standards, follow ethical practices, and comply with regulatory requirements.
10. Supply Chain Transparency:
- Blockchain Technology: Emerging technologies such as blockchain are being used to create a transparent and secure supply chain. Blockchain allows medical device manufacturers to track the origin and movement of materials and components throughout the supply chain in real-time, ensuring transparency and trust in the quality of materials.
- Real-Time Monitoring: Technologies such as RFID and IoT can enable real-time tracking of devices and materials as they move through the supply chain, providing better visibility and control.
11. Impact on Innovation and Product Development:
- Faster Time-to-Market: A well-structured supply chain reduces the time taken for components to be sourced and integrated into the final product, accelerating time-to-market for new medical devices.
- Collaboration for Innovation: Suppliers often play a key role in product innovation, working closely with manufacturers to provide new materials or components that enhance the functionality of medical devices. This close collaboration enhances the overall product development process.
12. Customer Satisfaction:
- Product Availability: Ensuring that medical devices are readily available when needed by healthcare providers is critical to patient care. A well-functioning supply chain guarantees that there is a consistent supply of devices, helping to avoid stockouts that could delay treatment or surgeries.
- After-Sales Support: The supply chain is also responsible for distribution and maintenance of medical devices post-sale. Efficient logistics, including spare parts availability and support services, contribute to customer satisfaction and device longevity.
The supply chain in medical device manufacturing is a complex, multifaceted system that directly impacts the efficiency, cost-effectiveness, and quality of medical devices. It plays an essential role in ensuring that devices meet rigorous regulatory standards, are available when needed, and are produced using the highest-quality materials. A well-managed, resilient supply chain also helps mitigate risks, foster innovation, and build trust with healthcare providers and patients. As the medical device industry continues to evolve with advances in technology and globalization, supply chain management will remain a cornerstone for success in this highly regulated and competitive sector.