What are the most common challenges in medical device prototyping?

Medical device prototyping is a critical step in the development process, translating concepts into tangible designs. However, it comes with several challenges that can impact the timeline, costs, and success of the final product. Here are the most common challenges:
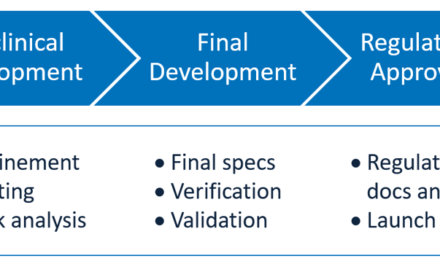
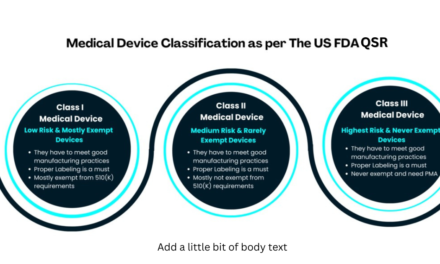
1. Balancing Innovation with Regulatory Compliance:
- Challenge: Prototypes must adhere to stringent regulatory standards (e.g., FDA, EU MDR) for safety, efficacy, and biocompatibility, even in the early stages.
- Impact: Balancing innovation and compliance can slow down the prototyping process and increase costs.
2. High Costs of Development:
- Challenge: Prototyping often requires expensive materials, advanced equipment, and specialized expertise.
- Impact: Budget constraints can limit the number of iterations, affecting the ability to refine the design.
3. Material Selection:
- Challenge: Identifying suitable materials that meet medical-grade standards while ensuring biocompatibility, durability, and manufacturability.
- Impact: Improper material choices can lead to prototype failures or non-compliance with regulatory requirements.
4. Precision and Miniaturization:
- Challenge: Many medical devices, such as implants or wearables, require high precision and miniaturized components.
- Impact: Achieving the necessary tolerances and functionality in a prototype can be technically challenging and resource-intensive.
5. Integration of Advanced Technologies:
- Challenge: Incorporating cutting-edge technologies like AI, IoT, or robotics into medical devices requires specialized expertise and tools.
- Impact: Integration can complicate the design process, leading to delays and increased costs.
6. Iterative Design Cycles:
- Challenge: Prototypes often require multiple iterations to address performance issues, usability, and regulatory feedback.
- Impact: Each iteration adds time and cost, especially if significant changes are needed.
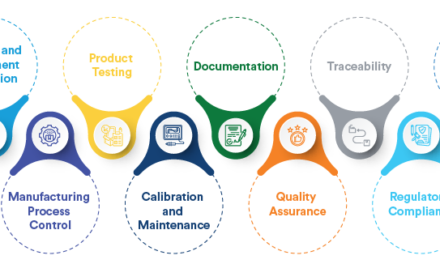
7. Testing and Validation:
- Challenge: Ensuring the prototype meets functional, safety, and usability requirements involves rigorous testing under various conditions.
- Impact: Failures in testing can lead to redesigns, extending timelines and increasing costs.
8. Biocompatibility and Sterilization:
- Challenge: For devices that come into contact with the human body, ensuring biocompatibility and the ability to withstand sterilization processes is critical.
- Impact: Selecting materials and manufacturing methods that meet these criteria can be complex.
9. Scalability to Manufacturing:
- Challenge: Prototypes often differ from mass-produced devices due to differences in materials, processes, or assembly techniques.
- Impact: A successful prototype may not translate easily to large-scale manufacturing, requiring further adjustments.
10. Time Constraints:
- Challenge: Accelerated development timelines, especially in competitive or urgent markets (e.g., during a pandemic), put pressure on the prototyping process.
- Impact: Time constraints can compromise design quality or lead to errors.
11. Collaboration Across Disciplines:
- Challenge: Prototyping involves collaboration among engineers, designers, medical professionals, and regulatory experts, each with different priorities.
- Impact: Miscommunication or conflicting goals can delay the process or result in suboptimal designs.
12. User-Centered Design:
- Challenge: Designing for ease of use, patient comfort, and clinical workflow compatibility often requires extensive feedback from end-users (patients and healthcare professionals).
- Impact: Iterative feedback cycles may add time and complexity.
13. Limited Access to Specialized Equipment:
- Challenge: Advanced prototyping often requires specialized tools like 3D printers, CNC machines, or cleanroom facilities, which may not be readily available.
- Impact: Limited access can slow down development or increase outsourcing costs.
14. Intellectual Property (IP) Concerns:
- Challenge: Protecting novel designs and technologies during the prototyping phase is critical to prevent infringement or loss of competitive advantage.
- Impact: IP protection processes can add administrative burdens and costs.
15. Incorporating Feedback:
- Challenge: Gathering and incorporating feedback from multiple stakeholders (e.g., doctors, patients, regulatory bodies) can be complex.
- Impact: Conflicting feedback may lead to design compromises.
16. Simulation vs. Real-World Testing:
- Challenge: Simulations and models may not always accurately predict real-world performance.
- Impact: Prototypes may perform differently in actual clinical settings, requiring further adjustments.
17. Environmental and Sustainability Factors:
- Challenge: Designing prototypes with sustainability in mind, such as using eco-friendly materials or minimizing waste, adds complexity.
- Impact: Balancing sustainability with functionality and compliance can be difficult.
18. Risk of Failure:
- Challenge: Prototypes may fail due to unforeseen design flaws, incorrect assumptions, or technical limitations.
- Impact: Failures necessitate redesigns, which consume time and resources.
Strategies to Overcome Challenges:
- Collaborative Development: Engage multidisciplinary teams early to align on goals and streamline decision-making.
- Rapid Prototyping Tools: Utilize technologies like 3D printing for faster iterations and cost efficiency.
- Material Optimization: Conduct thorough material research and testing to avoid costly redesigns.
- User Feedback Loops: Involve end-users from the beginning to ensure the design meets practical needs.
- Simulations and Modeling: Leverage advanced simulation tools to predict performance before physical prototyping.
- Scalability Planning: Design prototypes with scalability and manufacturability in mind from the outset.
Prototyping is a critical yet complex phase in medical device development. Addressing challenges like regulatory compliance, material selection, scalability, and testing requires a strategic approach, collaboration, and investment in advanced tools and processes. By tackling these challenges effectively, manufacturers can create prototypes that pave the way for successful and market-ready devices.